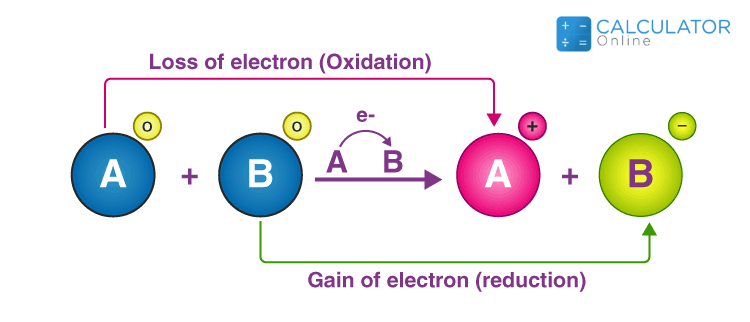
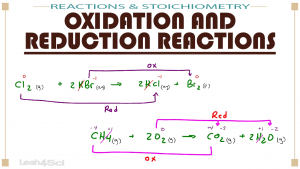
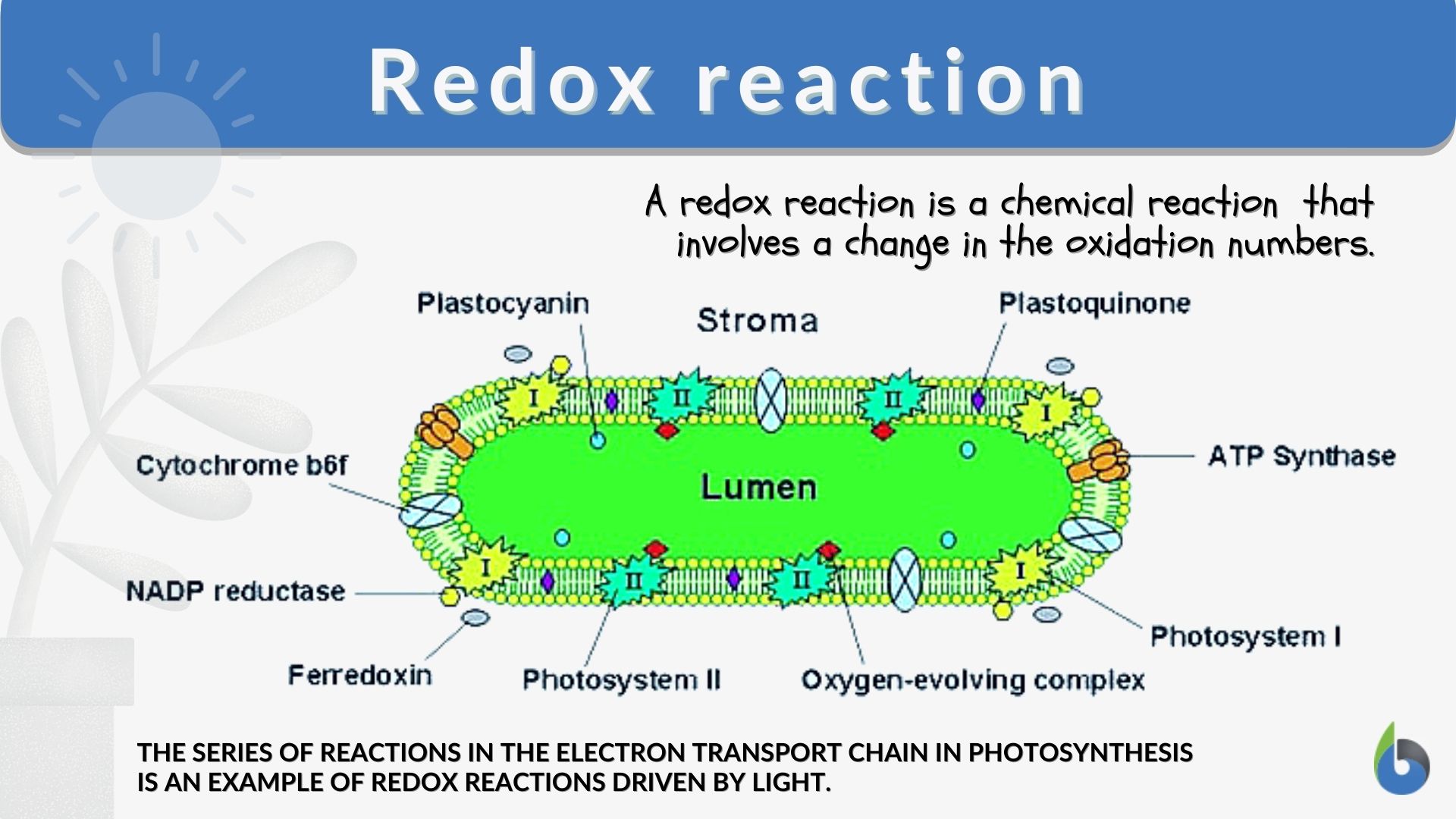
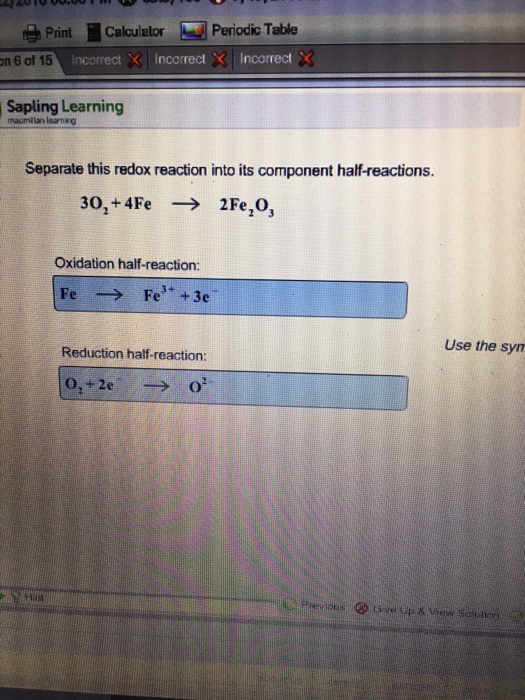
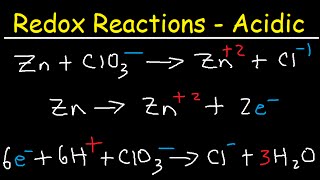SOLVED: Problems section You must show your work to get points Your calculator used for manual calculations only not for storage programming: 16) Consider the following unbalanced oxidation-reduction equation performed in basic
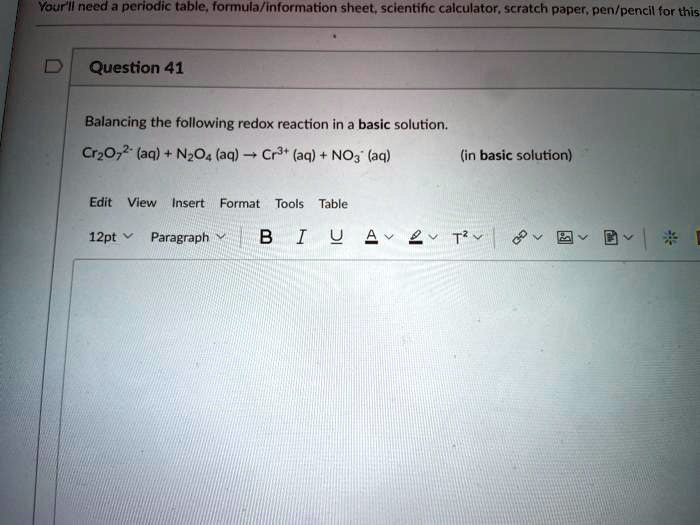
SOLVED: Vaurilneed periodic table, formula/ Information sheet; scientific calculator scratch paper; pen/pencil for this Question 41 Balancing the following redox reaction in a basic solution: Cr2Oz2* (aq) NzO4 (aq) Cr3t (aq) NOa" (

Redox Reactions Have you ever drank from an aluminum can? Ever used a flashlight? Use your calculator on a test recently? Enjoy exercising? - ppt download



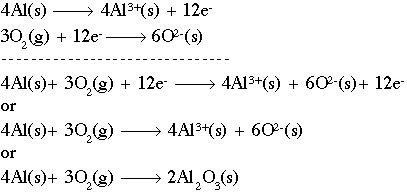


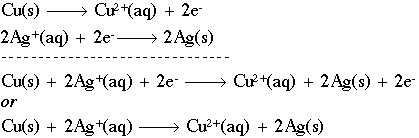
.png)