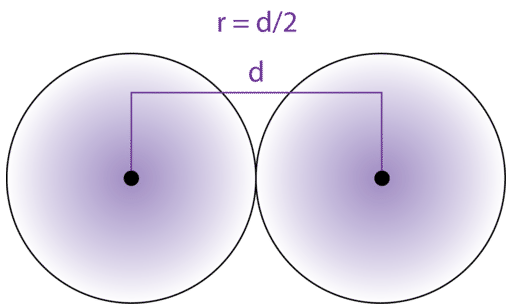
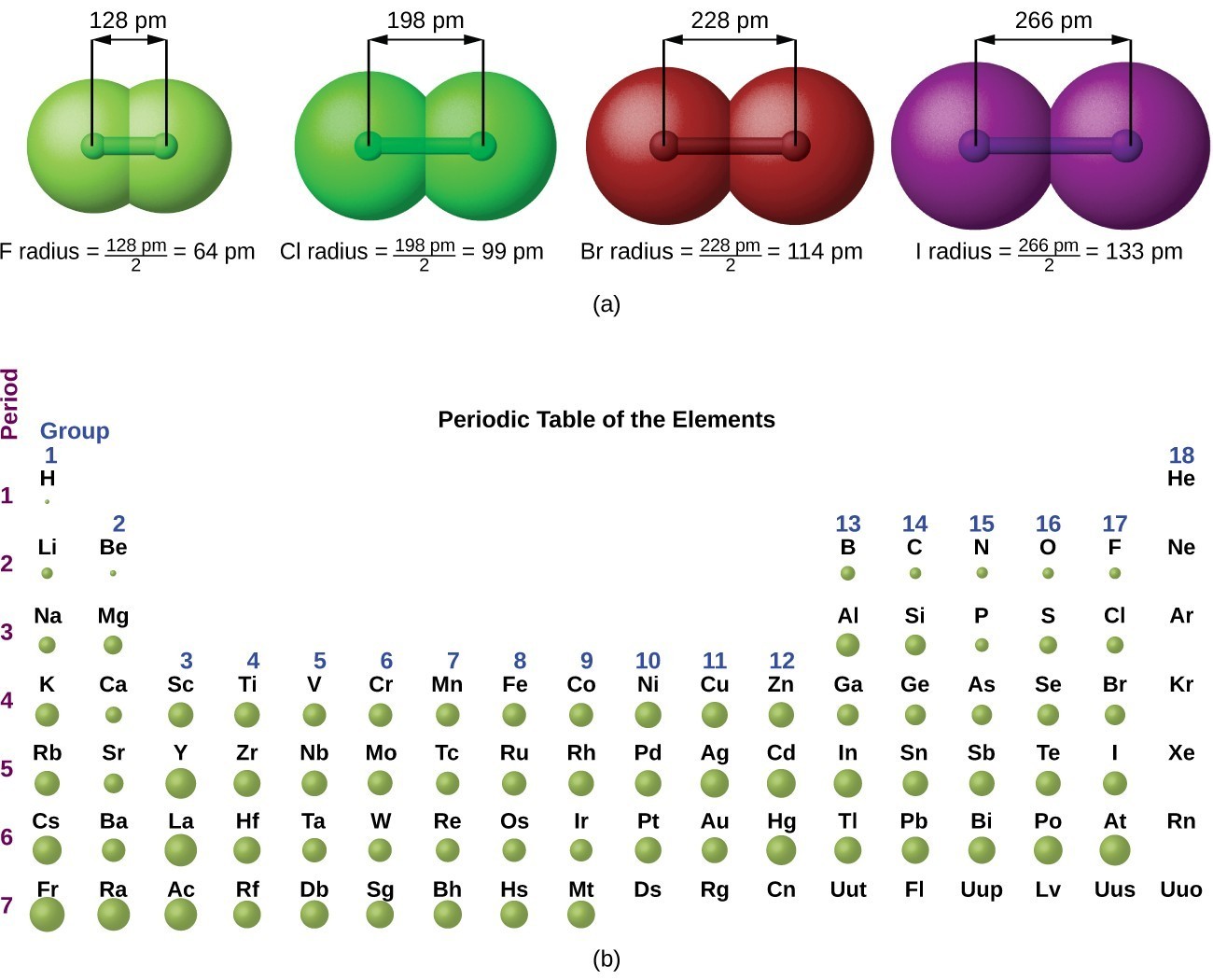
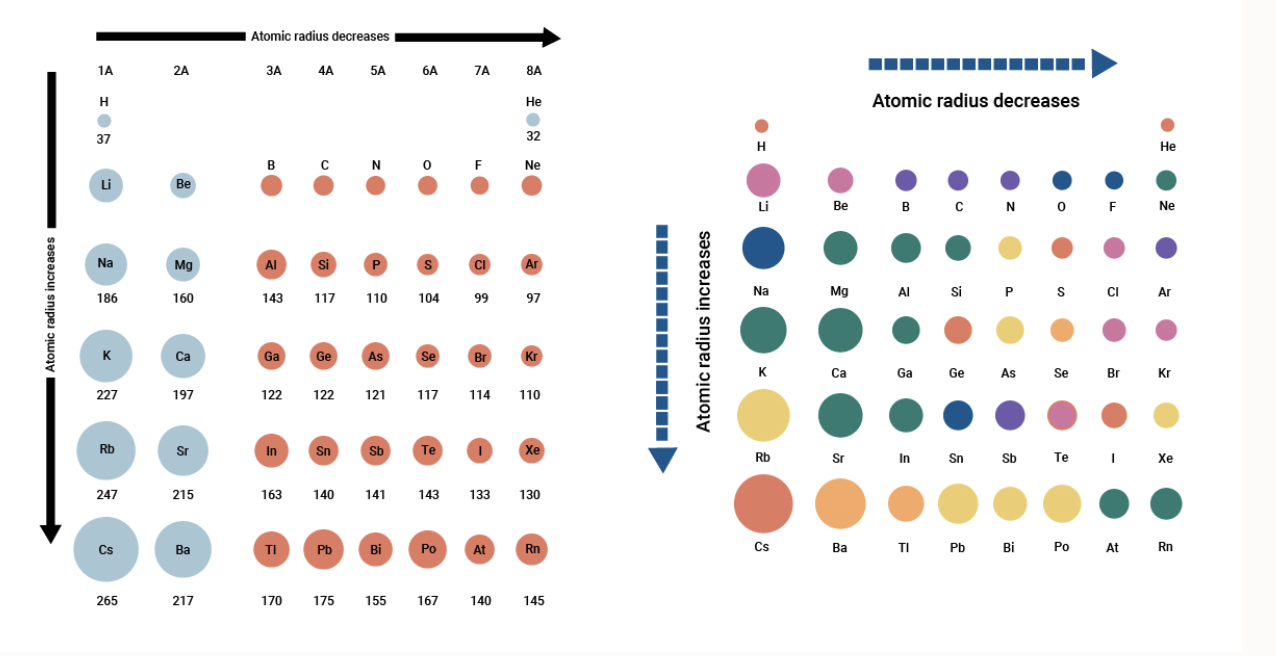
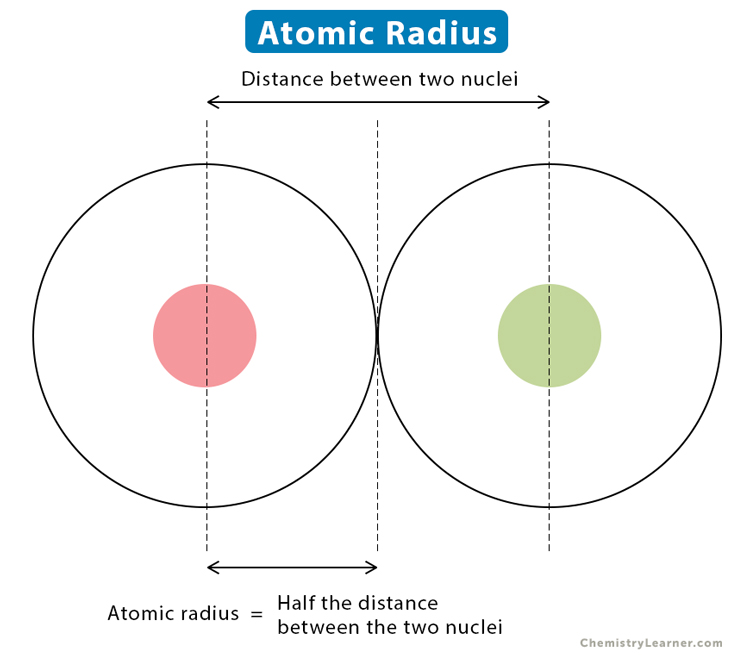
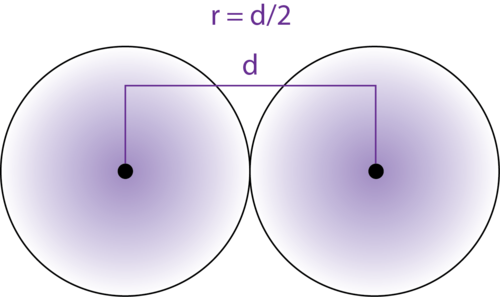
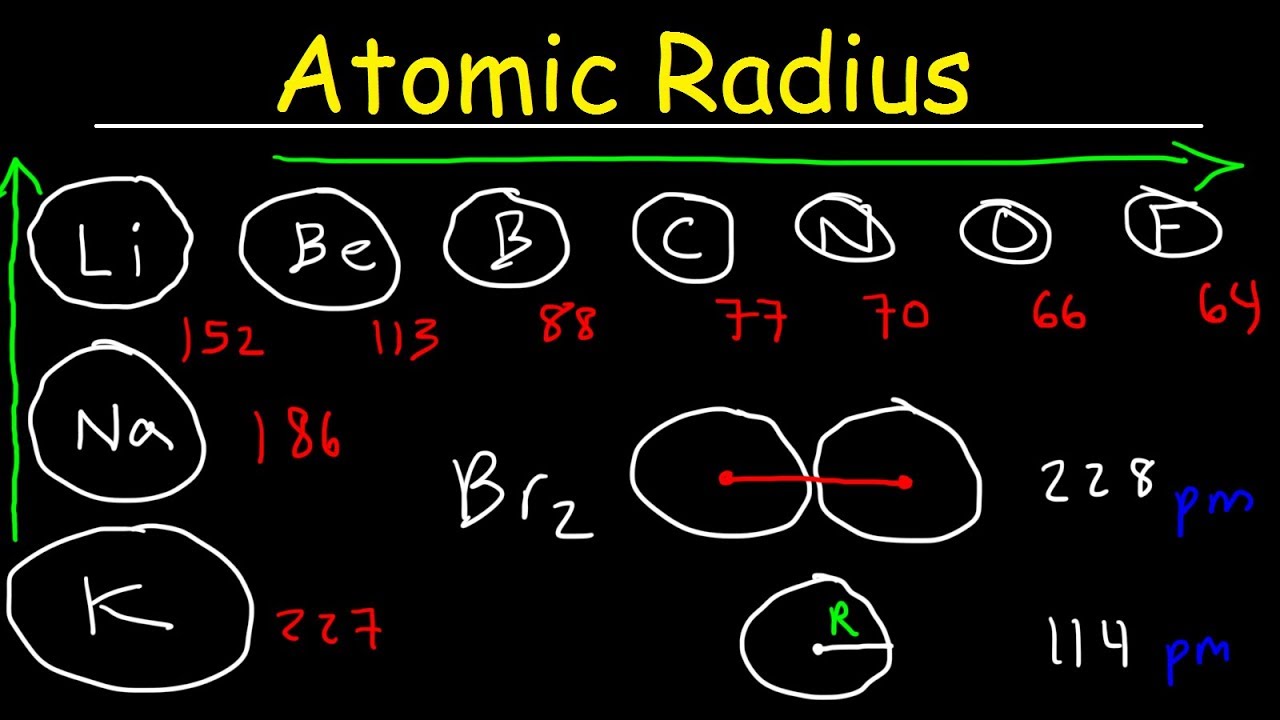
Welcome to Chem Zipper.com......: Atomic radius is the order of 10^-8 cm. and nuclear radius is the order of 10^--13 cm. Calculate what fraction of atom is occupied by nucleus

Atomic Radius Measurements of Diatomic Molecules Stock Vector - Illustration of hydrogen, material: 192485256

Calculate atomic radius of elementary silver which crystallites in face centered cubic lattice with unit - Brainly.in
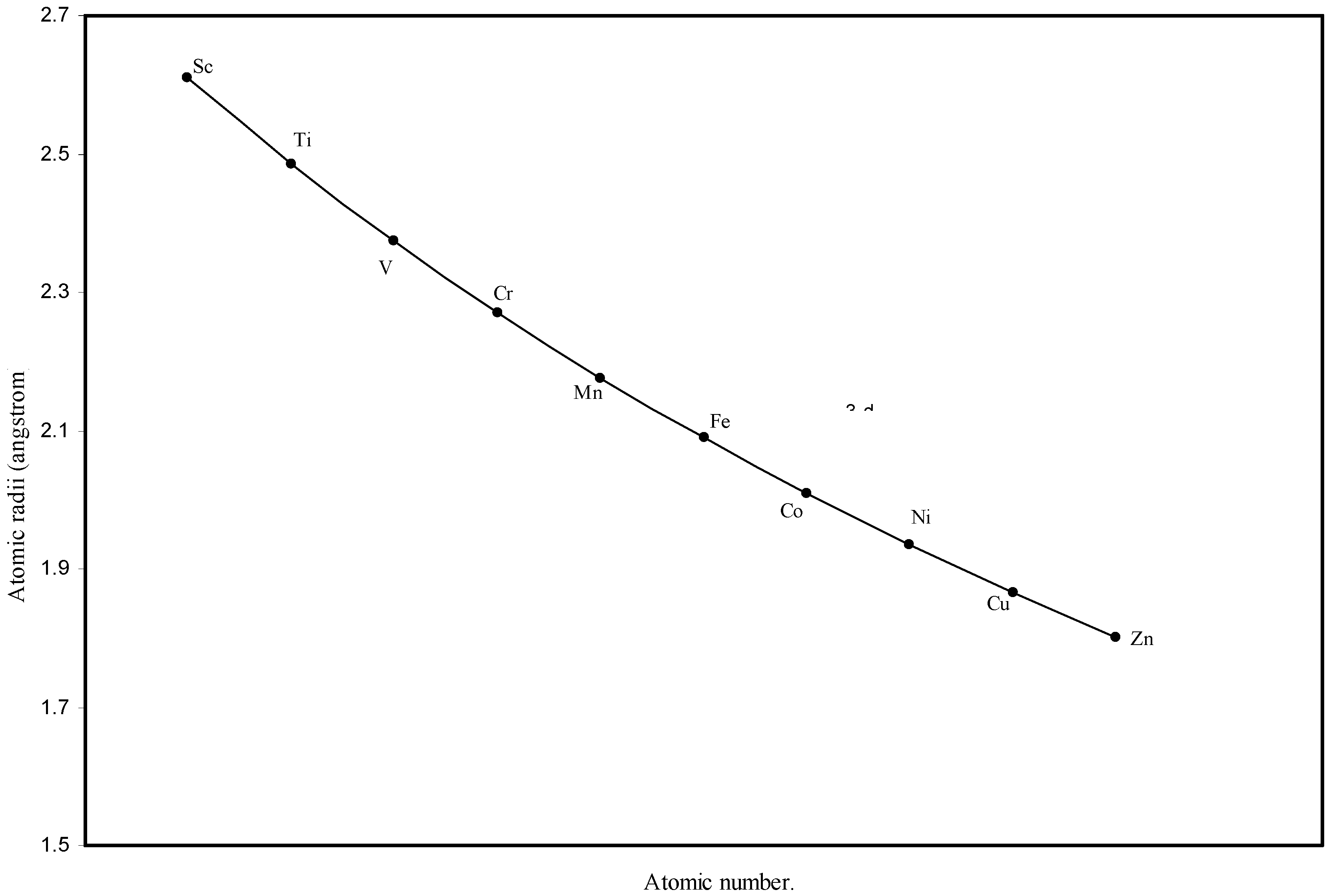
IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange
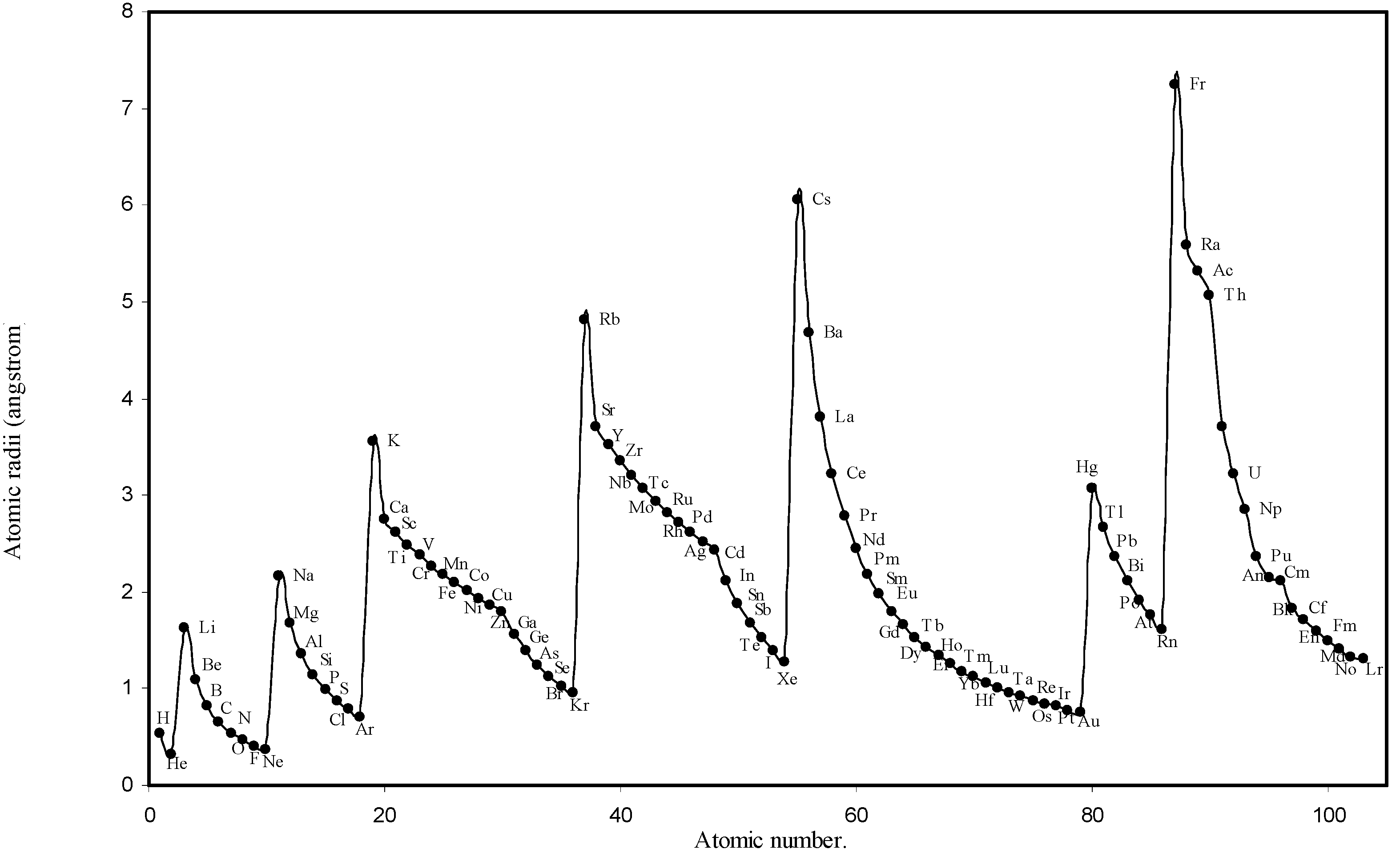
IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii

Atomic radius of `Li is 1.23 Å` and ionic radius of `Li^(+)` is `0.76 Å`. Calculate the percentage - YouTube