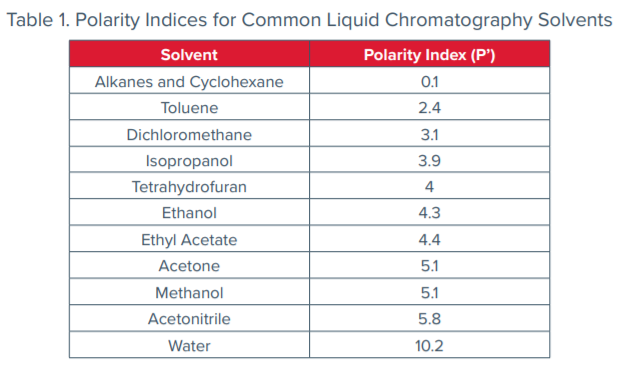
Solvent Polarity of Cyclic Ketone (Cyclopentanone, Cyclohexanone): Alcohol (Methanol, Ethanol) Renewable Mixed-Solvent Systems for Applications in Pharmaceutical and Chemical Processing | Industrial & Engineering Chemistry Research

Intermolecular forces – dipole – dipole forces Lesson Objectives: To describe the interaction of molecules by permanent dipole – dipole To compare dipole. - ppt download

Is cyclohexane an ionic, molecular nonpolar, or molecular polar compound? What intermolecular forces are present? | Homework.Study.com